Kossel-Lewis Approach to Chemical Bonding
Kossel-Lewis Approach to Chemical Bonding: Overview
This topic covers concepts such as Formal Charge, Applications of Formal Charges, Drawbacks of the Lewis Octet Rule, Lewis Acids, Odd Electron Molecules, Expanded Octet, Formation of Compounds by Noble Gases, Coordinate Bond, Chemical Compounds, etc.
Important Questions on Kossel-Lewis Approach to Chemical Bonding
The xenon reacts with oxygen forms its oxides with different composition of oxygen .
The xenon reacts with oxygen forms its oxides with different composition of oxygen _____.
_____ is the noble gas forms various fluoride compounds.
Xenon, a noble gas can form its fluorides.
Mention the fluoride compounds of the noble gases.
Give 2 drawbacks of Kossel's Postulate.
How will you calculate the valency of potassium?
Define the group valency.
List some odd electron molecules.
Identify odd-electron molecules from the following:
Which of the following oxides is an odd-electron molecule.
Which of the following bonds can be present in a chemical compound?
Hydrogen gas is a chemical compound.
Which of the following is a Chemical Compound?
Which of the following is not a Chemical Compound?
Bonding in this molecule can be understood to involve coordinate bonding.
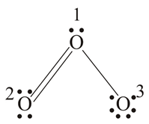
In the Lewis structure, the formal charge on the central atom of is
Aluminium chloride is a Lewis acid because it can donate electrons.
What are valence electrons? Write the number of valence electrons in the chlorine and carbon.